
Chemical Compositions
Mixture or "pure substance" ?

To a chemist, there is a fundamental distinction between a pure substance and a mixture.
But marketers, and through them, the general public, don't hesitate to describe a complex mixture such as peanut butter as "pure". Pure what?
But marketers, and through them, the general public, don't hesitate to describe a complex mixture such as peanut butter as "pure". Pure what?
Elements and compounds
2 HgO → 2 Hg + O2
... but the two products, metallic mercury and dioxygen, cannot be decomposed into simpler substances, so they must be elements.
Painting by Joseph Wright of Derby (1734-97) The Alchymist in Search of the Philosopher's Stone discovers Phosphorus
Elements and atoms

Formula and structure
The formula of a substance expresses the relative number of atoms of each element it contains. Because the formula can be determined by experiments on bulk matter, it is a macroscopic concept even though it is expressed in terms of atoms. What the ordinary chemical formula does not tell us is the order in which the component atoms are connected, whether they are grouped into discrete units (molecules) or are two- or three dimensional extended structures, as is the case with solids such as ordinary salt. The microscopic aspect of composition is structure, which in its greatest detail reveals the relative locations (in two or three dimensional space) of each atom within the minimum collection needed to define the structure of the substance.Macroscopic |
Microscopic |
|
| Substances are defined at the macroscopic level by their formulas or compositions, and at the microscopic level by their structures. | The elements hydrogen and oxygen combine to form a compound whose composition is expressed by the formula H2O. | 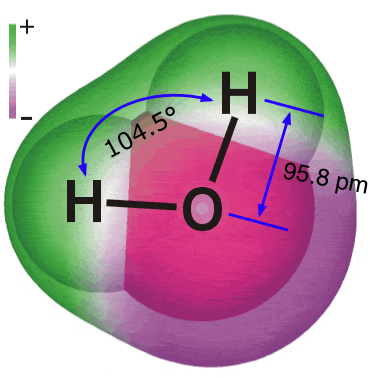 |
| Chemical substances that cannot be broken down into simpler ones are known as elements. The actual physical particles of which elements are composed are atoms or molecules. |  |
(This animation does not properly represent the actual vibrational motions of the molecule.)
|
Compounds and molecules
As we indicated above, a compound is a substance containing more than one element. Since the concept of an element is macroscopic and the distinction between elements and compounds was recognized long before the existence of physical atoms was accepted, the concept of a compound must also be a macroscopic one that makes no assumptions about the nature of the ultimate .
Thus when carbon burns in the presence of oxygen, the product
carbon dioxide can be shown by (macroscopic) weight measurements to
contain both of the original elements:
C + O2 → CO2
10.0 g + 26.7 g = 36.7 g
One of the important
characteristics of a compound is that the proportions by weight of each
element in a given compound are constant. For example, no matter what
weight of carbon dioxide we have, the percentage of carbon it contains
is (10.0 / 36.7) = 0.27, or 27%.
Molecules
A molecule is an assembly of atoms having a fixed composition, structure, and distinctive, measurable properties.
Molecules suddenly became real in 1905, when
Albert Einstein showed that Brownian motion, the irregular microscopic
movements of tiny pollen grains floating in water, could be directly
attributed to collisions with molecule-sized particles.
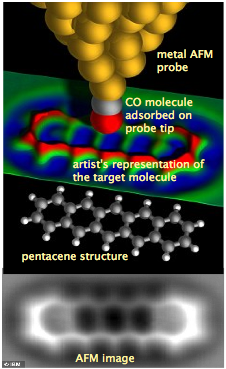
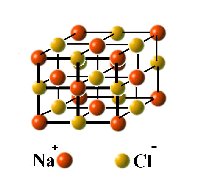
The atomic composition of a molecule is given by its formula. Thus the formulas CO, CH4, and O2 represent the molecules carbon monoxide, methane, and dioxygen. However, the fact that we can write a formula for a compound does not imply the existence of molecules having that composition. Gases and most liquids consist of molecules, but many solids exist as extended lattices of atoms or ions (electrically charged atoms or molecules.) For example, there is no such thing as a "molecule" of ordinary salt, NaCl (see below.)
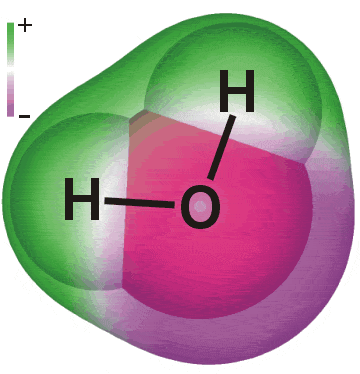
Confused about the distinction between molecules and compounds?
Maybe the following will help: |
 |
 |


 The
real importance of Chemistry is that it serves as the interface to
practically all of the other sciences, as well as to many other areas of
human endeavor. For this reason, Chemistry is often said (at least by
chemists!) to be the "central science".
The
real importance of Chemistry is that it serves as the interface to
practically all of the other sciences, as well as to many other areas of
human endeavor. For this reason, Chemistry is often said (at least by
chemists!) to be the "central science".  Do you remember the
Do you remember the 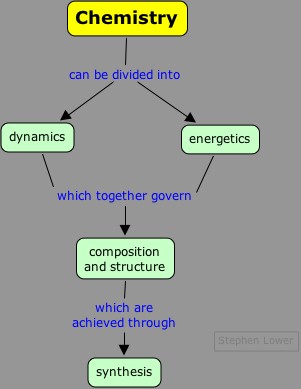 At the most fundmental level, chemistry can be organized along the lines shown here.
At the most fundmental level, chemistry can be organized along the lines shown here. 

.png)


